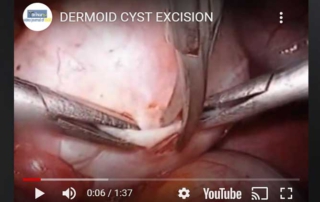
Excision of dermoid cyst
Information Authors: O. Sizzi, A. Rossetti The video shows the technique for the removal of a dermoid cyst. The [...]
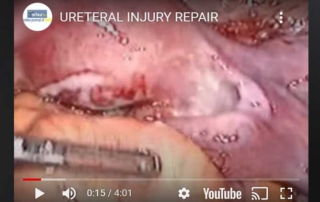
Ureteral injury repair
Information Authors: A.Rossetti, O.Sizzi Ureteral injury is one of the possible complications of endometriosis surgery. Ureteral dissection can cause [...]
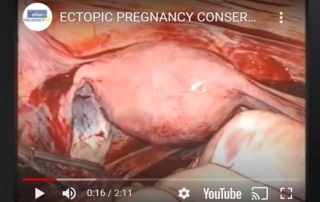
Conservative management of ectopic pregnancy
Information Authors: O. Sizzi, A. Rossetti The ectopic pregnancy is a possible life threatening condition. Only if the beta-HCG [...]
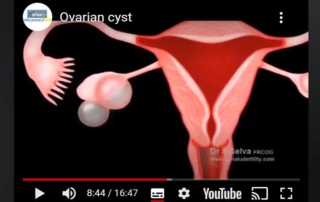
Ovarian cyst
Information Authors: Dr. S. Selva This is a patient education video on ovarian cysts. Video [...]
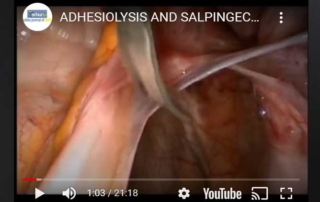
Adhesiolysis and salpingectomy for endometriosis
Information Authors: O. Sizzi, Alfonso Rossetti The video shows the case of a patient who had had a previous [...]
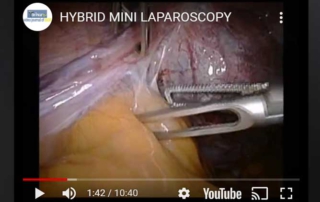
Hybrid mini laparoscopy salpingectomy
Information Authors: O. Sizzi, A. Rossetti Minilaparoscopy surgery usually uses 3 or 2 mm instruments. The benefits of this [...]
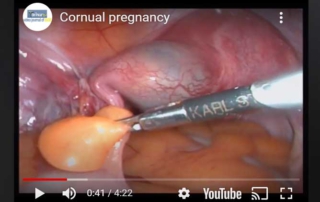
Cornual pregnancy
Information Authors: Recai Pabuccu 1 Prof. M.D. , Mine Kiseli 1 M.D. , Aslı Yarcı Gursoy 1 M.D. , [...]
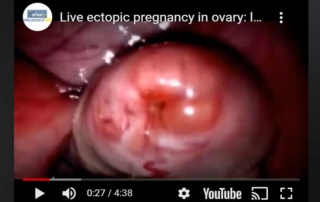
Live ectopic pregnancy in ovary: laparoscopic management
Information Authors: Dr Meenu Agarwal Live ectopic pregnancy in ovary: laparoscopic management. The video presents a case of [...]
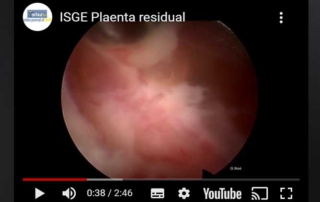
Removal of placental residual
Information Authors: GUNTER NOE The video shows the hysteroscopic removal of placental residual after a courrettage. The removal was [...]
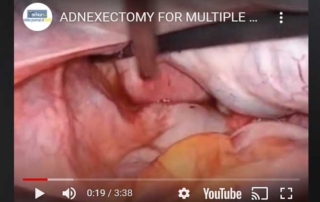
Adnexectomy for multiple mucinous cysts
Information Authors: A. Rossetti, O. Sizzi This video shows the technique of adnexectomy. The patient was affected by multiple [...]
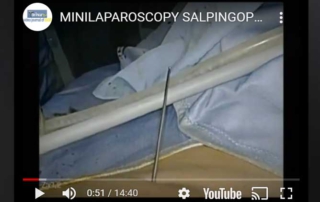
Hybrid minilaparoscopy: salpingoplasty
Information Hybrid minilaparoscopy: salpingoplasty. Video
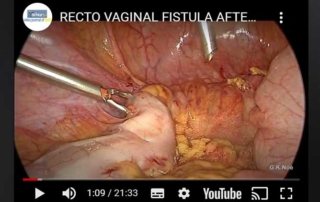
Recto vaginal fistula after supracervical hysterectomy
Information Authors: Guenter K. Noé MD Phd Medical Director Communal Hospital of Dormagen Head of Department OB/Gyn Ass. Professor [...]